|
Fewer patients proceeding to a subsequent line of treatment in real-world could partly explain this.įor patients with stage IV non-small cell lung cancer (NSCLC), immune checkpoint inhibitors (ICIs) have demonstrated promising results in disease progression and survival in randomized clinical trials (RCTs) 1, 2, 3, 4, 5, 6. 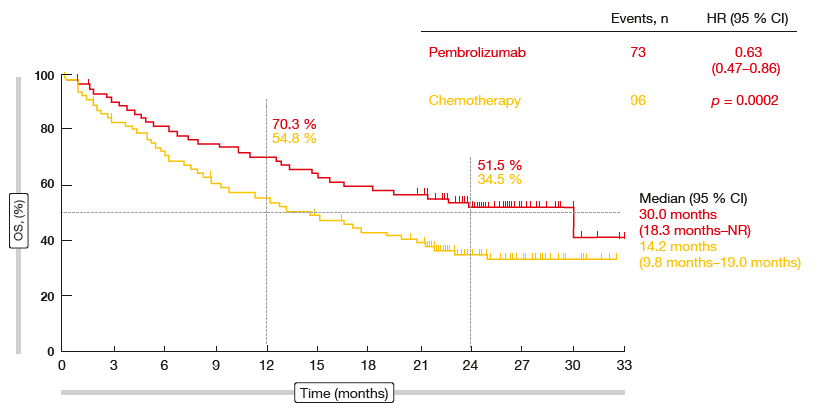
However, there is a gap in OS for 1L pembrolizumab. There is no EE gap for PFS from immunotherapy in patients with stage IV NSCLC. Receiving subsequent lines of treatment was less frequent in real-world compared to trials. OS was significantly shorter in real-world for 1L pembrolizumab (HR 1.55 95% CI 1.07–2.25). For both settings, PFS times were comparable between real-world and trials (HR 1.08 (95% CI 0.75–1.55), and HR 0.91 (95% CI 0.74–1.14), respectively). Nivolumab was started as 2L treatment in 141 patients. From 1950 diagnosed patients, 1005 (52%) started with any 1L treatment, of which 83 received pembrolizumab. Progression-free survival (PFS) and overall survival (OS) from first-line (1L) pembrolizumab and second-line (2L) nivolumab were compared with clinical trial data by calculating hazard ratios (HRs). All patients diagnosed with stage IV non-small cell lung cancer (NSCLC) in 2015–2018 in six Dutch large teaching hospitals (Santeon network) were identified and followed-up from date of diagnosis until death or end of data collection.  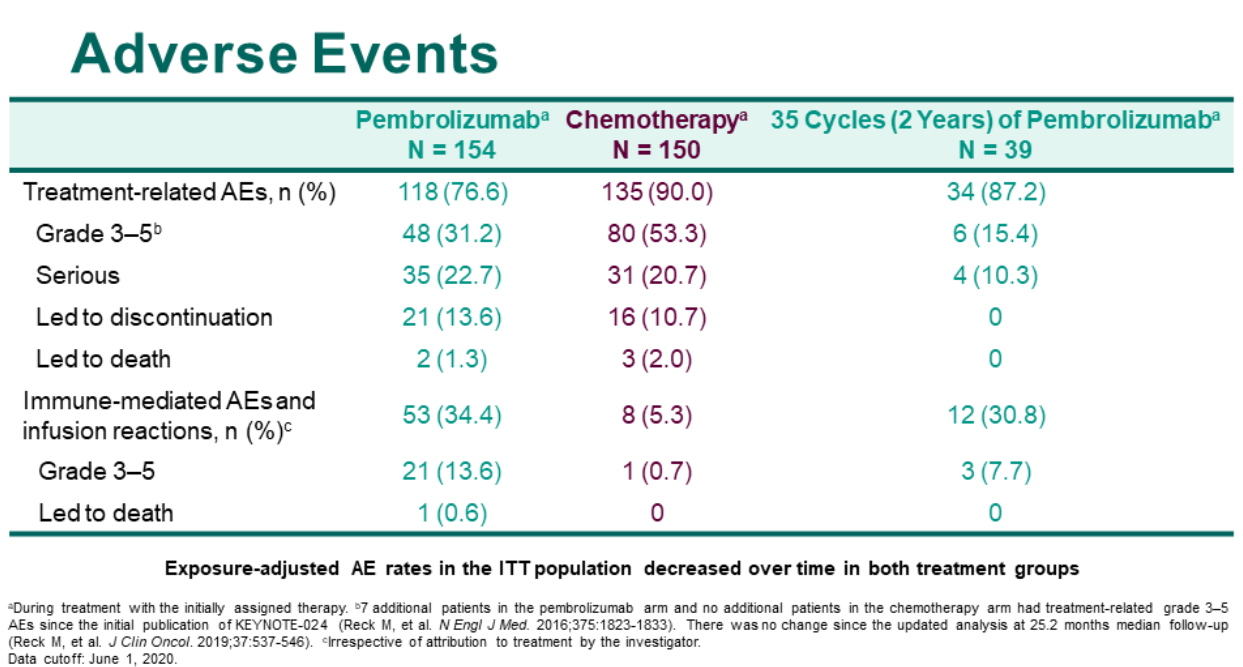
This study aims to assess how clinical outcomes of immunotherapy in real-world (effectiveness) correspond to outcomes in clinical trials (efficacy) and to look into factors that might explain an efficacy-effectiveness (EE) gap.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
